- Home
- About
- Contact
- La haine youtube
- What can i do with the amazon fire hack
- Emma stone sleeping dogs
- Yoga undressed advanced video
- Mnemonic for hydrophobic amino acids
- Kgf kannada movie release date
- Simpsons hit and run iso gamecube
- White heart baekgu
- Hard reset iphone 8 plus without itunes
- Visual certexam software
- Uptown girl billy joel mp3 download
- Who is yogini liz from yoga undressed
- Cisco wlc delete snmp trap receiver cli
- Top pc games under 1gb download
- Vb net 2010 app config connectionstrings
- Resident evil 6 pc walkthrough
- Vb net 2010 time triggered event
- Drastic ds emulator r2-5-0-3a obb
- Uncharted 3 game of the year edition or regular
- Moddb sven coop player models
- Project x love potion disaster game free
- Come a little closer bass tab
- Wii dance michael jackson thriller
- Indian idol naina thag lenge
- Astr bleeding love
- Hot bhojpuri video song 2016
- Futura font family vk
- Need for speed most wanted maps
The complexity of a protein structure is determined by its sequence of amino acids and the chemical nature of their variable group side chains. But then you have your workers, transportation system and the very structure of the cell city, which are all made of proteins- which in turn are made of amino acids. I like to think of the cell as a self contained city where the nucleus is the capital, the mitochondria is the power plant and so on. Long chains of amino acids make up proteins, which in turn make up many structural and functional cell components. Available here Image Courtesy:ġ.”Amino acids”By Dan Cojocar (CC BY-SA 3.0) via Commons WikimediaĢ.Amino acids are the building blocks of living things. “Amino Acid.” Encyclopædia Britannica, Encyclopædia Britannica, Inc., 30 Oct. The key difference between hydrophobic and hydrophilic amino acids is that the hydrophobic amino acids are nonpolar whereas the hydrophilic amino acids are polar. Moreover, according to the polarity, there are two types as hydrophilic and hydrophobic amino acids. In brief, amino acids are the building blocks of proteins. Summary – Hydrophobic vs Hydrophilic Amino Acids The below illustration shows detailed description on the difference between hydrophobic and hydrophilic amino acids. As another important difference between hydrophobic and hydrophilic amino acids, hydrophobic ones occur at the centre of proteins while hydrophilic amino acids are on the surface. Moreover, hydrophobic amino acids have long side chains with mostly carbon and hydrogen atoms whereas hydrophilic amino acids have either short side chains or side chain with hydrophilic groups. Hence, this is the key difference between hydrophobic and hydrophilic amino acids. Hydrophobic amino acids are a type of amino acids which have a nonpolar nature while hydrophilic amino acids are a type of amino acids in which have a polar nature. What is the Difference Between Hydrophobic and Hydrophilic Amino Acids? Since water is a polar solvent and these amino acids are also polar, they can dissolve in water. The name “hydrophilic” derives because it attracts water.
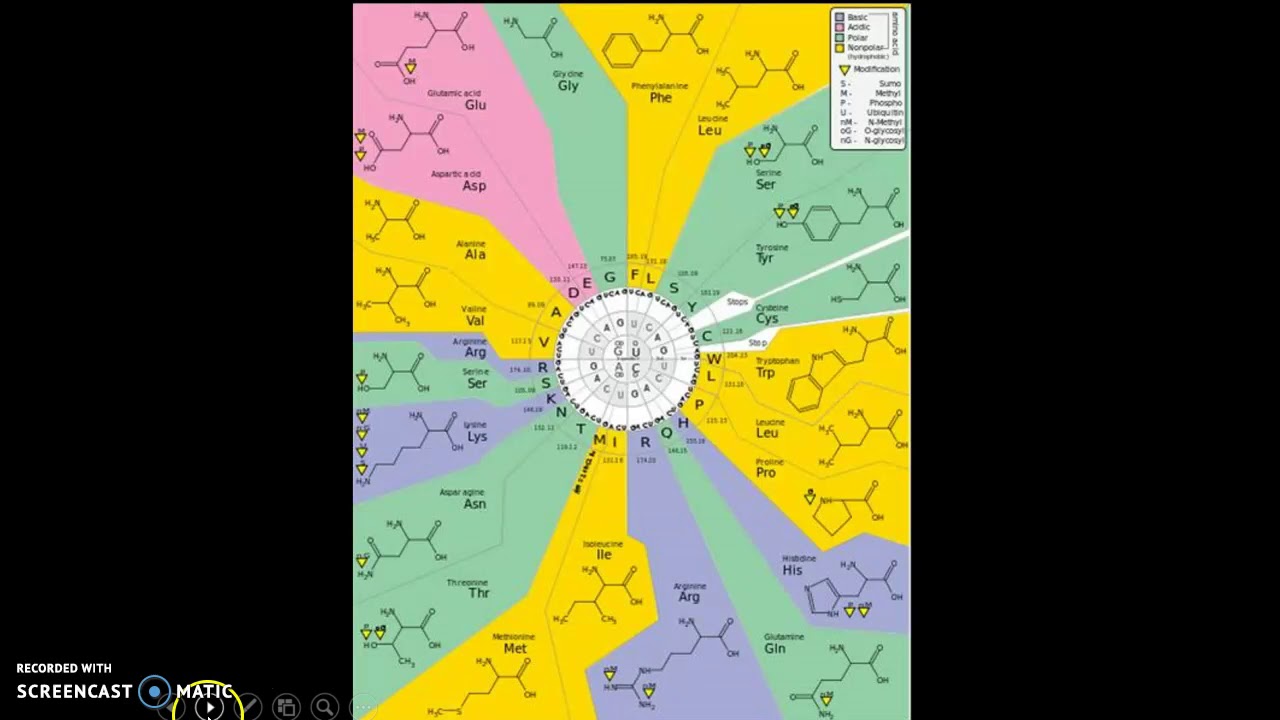
Hydrophilic amino acids are a type of amino acids with a polar nature. Moreover, the hydrophobic amino acids among essential amino acids are as follows.

Therefore, they tend to repel from the water. Furthermore, they have small dipole moments. Thus, if the side chain is very long and consists mostly of carbon and hydrogen atoms, they are hydrophobic. This R group can be simply an atom (hydrogen atom) or a long side chain. An amino acid has the general formula in which a central carbon atom is attached with a hydrogen atom, a carboxyl group, an amine group and a side group (R group). Hence, the hydrophobic nature of these compounds arises due to the side chains they have in their chemical structure. Since these amino acids are nonpolar, they cannot dissolve in water. Likewise, the name “hydrophobic” derives because it does not interact with water (“hydro” – water). Hydrophobic amino acids are a type of amino acids with a nonpolar nature. Summary What are Hydrophobic Amino Acids? Side by Side Comparison – Hydrophobic vs Hydrophilic Amino Acids in Tabular Formĥ. They are different from each other mainly based on the polarity. Moreover, we can categorize them as hydrophilic and hydrophobic amino acids, depending on their physicochemical nature.

Furthermore, amino acids are mainly in two types as essential and nonessential amino acids. A protein is a giant polymer molecule which is an essential component of all living organisms. The key difference between hydrophobic and hydrophilic amino acids is that the hydrophobic amino acids are nonpolar whereas the hydrophilic amino acids are polar.Īmino acids are the building blocks of proteins.
